This unique program provides objective post-operative arrhythmia analysis that dramatically improves a physician’s ability to assess the clinical outcomes from these complex procedures. Cardiac Centers (US News and World Report), utilize the LifeStar ACT System in their monitoring programs, while over 20 key cardiac institutions have enrolled their AF catheter and surgical ablation patients in the first-to-market LifeWatch AF Patient Care Program. This exceptional growth is directly attributed to the successful adoption of the ACT monitoring system in hospitals, cardiology practices, and electrophysiology labs. 
LifeStar ACT, which was launched in January of 2007, is the catalyst for the rapid growth in LifeWatch’s revenues. Kopelman is a highly esteemed electrophysiologist who has authored and co-authored a number of papers in highly referred publications and serves as the Chairman of the LifeWatch Medical Advisory Board. Kopelman, Director of the Cardiac Electrophysiology Laboratory at the Fuqua Heart Center & the Piedmont Heart Institute. Disease state management requires more accurate diagnostic tools such as the ACT III to allow for early detection of a worsening condition in patients that are minimally symptomatic” according to Dr. 
“In my opinion, real-time outpatient cardiac monitoring using this new and unique 3-channel configuration will extend physicians management of patient conditions which have been traditionally monitored in an inpatient setting. The sophisticated algorithm and advanced wireless and web-based technologies of ACT III enhances data collection, analysis, transmission and reporting, and can be utilized in standard and alternative patient monitoring programs, such as Specialized Atrial Fibrillation (AF) Patient Care and Congestive Heart Failure (CHF) programs. 
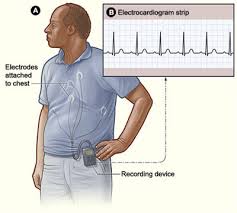
This ensures that no valuable data is lost if a patient is disconnected from the cellular phone. The device has 6 hours of memory in the sensor and a flash memory of up to 21 days of data on the ACT Cell phone monitor. With up to 21-days of real-time ECG monitoring and the provision of ST deviation analysis, the ACT III provides more sensitive and specific data for initial or early detection of arrhythmia in patients that have limited or atypical symptoms. The ACT III monitor offers broader functionality with its high-performance multi-channel Ambulatory Cardiac Telemetry in one simple-to-use system. Higher sensitivity facilitates expansion into alternative patient monitoring markets The ACT III monitor, which requires only minimal patient intervention, significantly improves patient compliance and increases diagnostic yield. This call center is staffed by certified cardiac technicians, who interpret the data and initiate physician notification based on pre-determined criteria. The ACT III monitor automatically detects and sends heart rhythm abnormalities to a 24/7 LifeWatch monitoring call center. This advanced monitoring device, cleared by the FDA in June 2008, provides enhanced heart arrhythmia detection through its 3-channel ECG functionality and ST deviation analysis that can measure ischemic changes. announced today that LifeWatch has launched its next-generation LifeStar ACT III Platinum (ACT III) monitor in the United States. Card Guard AG (SWX Swiss Exchange: CARDG), the parent company of LifeWatch Corp.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
